There has been an intense focus on the development of an effective vaccination since the World Health Organization (WHO) classified COVID-19 a pandemic in March 2020.
- When Can Baby Sleep With Blanket? Comprehensive Guide Update 05/2026
- How To Wash Pillows? Helpful Tips To Remember Update 05/2026
- How to Stop Mattress Topper from Sliding? Best Guide Update 05/2026
- How Do Different Addictions Affect Sleep? 5 Questions for Your Doctor Update 05/2026
- How Is Sleep Quality Calculated? Perfect Information For You Update 05/2026
Three separate vaccines have been given emergency use authorisation by the Food and Drug Administration (FDA) in the United States, and two of them have already obtained full FDA approval for those over 16. More than 87 percent of individuals in the United States had received at least one dose of the COVID-19 vaccination as of mid-December 2021.
Bạn đang xem: What Is the Relationship Between COVID-19 Vaccines and Sleep? Update 05/2026
Because of the importance of COVID-19 vaccinations to public health, it is critical that the public has easy access to reliable, scientifically sound information regarding these vaccines.
What Is COVID-19?
A novel coronavirus known as SARS-CoV-2 was discovered in Wuhan, China, in late 2019. COVID-19 is the term given to the sickness that can develop from the virus.
A cough or sneeze can spread the SARS-CoV-2 virus to anyone within six feet of each other if they are in close proximity to each other.
People who are more than six feet apart can also spread the virus by airborne transmission.
It’s possible to inhale or exhale aerosols that contain the virus and spread the disease through the air. In confined spaces with inadequate ventilation, virus propagation is more likely.
Touching infected surfaces is a less common means of spreading the coronavirus.
Patients infected with COVID-19 may experience a wide range of symptoms. Other symptoms include fever, coughing, difficulty breathing, a loss of taste and/or smell, a headache and/or body ache and nausea or vomiting as well as a runny or congested nose.
COVID-19 can cause difficulty breathing, pain or pressure in the chest, confusion, inability to wake up or stay awake, and pale/discolored skin, lips, or nail beds in really severe forms of the disease. See a doctor right away if you have any of these symptoms.
Asymptomatic and presymptomatic people can still spread the virus, even if they don’t display any symptoms yet.
Multiple coronavirus variants have arisen since the outbreak of COVID-19. In comparison to the original SARS-CoV-2 virus, many of these variations, such as Alpha and Omicron, can be propagated more quickly and easily than the original (B.1.1.7).
What COVID-19 Vaccines Are Available?
The Pfizer-BioNTech COVID-19 Vaccine, marketed as Comirnaty by Pfizer-BioNTech, received FDA approval for use in adults 16 and older on August 23, 2021. For people over the age of 18, the FDA has given full approval to the Spikevax vaccine, previously known as the Moderna COVID-19 Vaccine. The vaccine’s approval by the FDA was based on considerable data showing its safety and efficacy.
The FDA has approved the use of the Johnson & Johnson (Janssen) COVID-19 Vaccine for people of specific ages with an emergency use authorisation (EUA). The emergency use of the Pfizer-BioNTech vaccine for children ages 5 to 15 has not been revoked.
| Vaccine Name | Type of Vaccine | Who Can Get This Vaccine? | Required Doses | When Are You Fully Vaccinated? | Authorized Booster Shots | Date of Initial FDA Authorization |
|---|---|---|---|---|---|---|
| Pfizer-BioNTech (Comirnaty) | mRNA | 5 years and older | 2 shots given 3 weeks apart (21 days) | 2 weeks after your second shot | Single booster for anyone 12 and over | December 11, 2020 |
| Moderna (Spikevax) | mRNA | 18 years and older | 2 shots given 4 weeks apart (28 days) | 2 weeks after your second shot | Single booster for anyone 18 and over | December 18, 2020 |
| Janssen (Johnson & Johnson) | Viral vector | 18 years and older | 1 shot | 2 weeks after your shot | Single booster for anyone 18 and over whose first shot was at least 2 months prior | February 27, 2021 |
All three of these vaccines are injected into a muscle in the upper arm and are delivered this way.
Clinical investigations involving thousands of adults and adolescents have proven their efficacy. When vaccines were given to patients in the studies, they were less likely to become sick, have severe symptoms, be hospitalized for treatment, or die of COVID-19. When compared to a placebo, the vaccinations provided superior protection with fewer side effects than expected.
Pfizer and Moderna have created mRNA vaccines. The immune system is taught to recognize and attack the virus through the use of a synthetic, non-lethal piece of the virus’ genetic material.
Janssen, part of Johnson & Johnson, developed a viral vector vaccine for the corporation. A genomic segment of SARS-CoV-2 is incorporated into a deactivated common cold virus in order to create a vaccine. An improved immune system can better identify and combat the coronavirus as a result of this.
Pfizer and Moderna vaccines require two doses to be adequately immunized in the majority of patients. The Food and Drug Administration has allowed a third dosage of these mRNA vaccines as part of the main vaccination series for persons with extremely impaired immune systems, such as organ transplant recipients.
The Food and Drug Administration (FDA) has approved booster doses for all adults 18 and older who have previously received a full vaccination schedule.
- All adults who were initially immunized with the Pfizer or Moderna vaccines are permitted to receive a booster shot. As a rule of thumb, this booster is given at least five months following the original vaccine.
- Anyone over the age of 18 who received their first shot during the previous two months and was initially immunized with the Johnson & Johnson vaccine is eligible for a booster shot.
Those above the age of 12 who have been completely vaccinated for at least six months are also eligible for the Pfizer vaccination booster.

Xem thêm : What Is Shift Work? Tips for managing shift work schedules Update 05/2026
The FDA has permitted a “mix-and-match” approach to booster doses, allowing any of the three authorized vaccines to be used. Booster shots may be administered with the same vaccine.
What COVID-19 Vaccines Are Under Development?
There are now three vaccines on the market in the United States, but more are being developed. If research studies show that they are safe and effective, they may be approved.
Vaccines developed by AstraZeneca have been approved by the European Medicines Agency but not the Food and Drug Administration (FDA). While the FDA is still reviewing a protein subunit vaccine created by Novavax, it has been authorized for use in Europe. A coronavirus protein subunit vaccination boosts the immune system’s ability to recognize and destroy the virus by adding the tiny protein into the vaccine.
As additional SARS-CoV-2 strains emerge, it is possible that improved booster vaccinations will be developed to combat a wider variety of coronavirus variations.
Research is also underway to develop universal coronavirus vaccines, which would protect against practically any current or future coronaviruses, as well.
What Is the Timeline for Vaccination?
Since the first COVID-19 vaccine was approved by the FDA in December 2020, vaccination has been ongoing. Federal administration oversees vaccine distribution, but states and local governments are generally responsible for administering vaccination programs.
The Pfizer vaccine was just made available to children aged 5 to 11 and to those aged 12 to 15 in all fifty states. Pfizer has also announced that it will seek emergency use authorization for its vaccine in children between the ages of six months and five years.. It is still being tested in a younger demographic by Moderna.
How Can I Get a COVID-19 Vaccine?
There are already COVID-19 vaccines accessible to all clinically recommended age groups in the United States (see table for details).
Contact your state and local health departments to find out where you can get vaccinated. In addition, you can seek advice from your physician.
In collaboration with the CDC, a Vaccine Finder website has been created to give data on vaccine availability across the country. It is possible to get a vaccine at a variety of locations such as health clinics and medical offices, hospitals, pharmacies, and mass vaccination sites.
It’s vital to remember that you won’t be able to pick and choose whatever vaccine brand you get. As a result, many vaccination locations have begun reporting the vaccine type and manufacturer. It’s always advisable to talk to your doctor if you have any questions or concerns regarding a particular immunization.
Are COVID-19 Vaccines Safe?
More than 535 million doses of the COVID-19 vaccine have been administered in the United States since December 2020, demonstrating the vaccine’s safety in studies involving tens of thousands of patients in several nations.
Pain or swelling in the arm where the injection was given, fever, chills, and headache are among the most common side effects. The Pfizer, Moderna, and Johnson & Johnson vaccines all cause substantial tiredness or exhaustion in many people.
After immunization, these side effects are usually gone within a few days and pose no long-term issues. Side effects are possible with either of the two doses of the Pfizer and Moderna vaccines, although the second dose has been the most frequently reported.
There have only been a handful of reports of serious adverse responses for all three immunizations. After receiving the shot, you will typically be requested to remain at the vaccination site for 15 minutes to check for any adverse reactions. This is the best moment to report any symptoms of an allergic response, such as facial swelling or rash, difficulty breathing or dizziness.
Women under the age of 50 have an increased chance of developing thrombosis with thrombocytopenia syndrome (TTS) after receiving the Johnson & Johnson vaccination. Fewer than 60 of the nearly 17 million people who have received the vaccine have experienced this side effect, which is extremely rare. The FDA and CDC have decided that the vaccine’s advantages outweigh its risks, so it can be used again. TTS has not been connected to any other approved vaccines.
Pre-Vaccination Precautions
Some care must be taken when receiving the COVID-19 vaccinations. If you have any of the following conditions:
- Have been diagnosed with an allergy.
- I’m currently suffering from a high temperature.
- Diagnosed or recently exposed to a person with COVID-19 symptoms.
- A blood thinner is being taken by them.
- I suffer from a condition that causes me to bleed.
- Are unable to fight infection.
- Don’t forget to take immunosuppressive medication.
- Do you have a baby on the way?
- Do you have a baby?
- Heart muscle or heart lining inflammation is possible.
- Received a previous dose of COVID-19 vaccination.
In order to guarantee a safe vaccination delivery, it is important to discuss these factors with your physician before getting vaccinated.
Expect to get a booklet outlining possible side effects when you come for your vaccination appointment. Medical experts at the vaccination site will be happy to answer any questions or concerns you may have about this form.
Who Shouldn’t Get the COVID-19 Vaccine?
Xem thêm : How Long Should A Mattress Last? Factors Influencing Mattress Lifespan Update 05/2026
Immunization should not be given to those who have previously developed an adverse reaction to a previous dose of the COVID-19 vaccine or to any of the vaccine ingredients.
How To Stay Safe After Vaccination
The coronavirus vaccine does not provide instant protection. Your body’s immune system takes some time to establish a reaction to COVID-19, and during this period, you are still vulnerable. While the vaccines have been proven to protect against serious sickness, it is not yet clear if they prevent the virus from being transmitted.
Immunization against COVID-19 significantly reduces the risk of severe illness, hospitalization, and mortality in those who have been vaccinated. As a result, they also have some immunity against disease. Vaccinated people, on the other hand, are still at risk of contracting COVID-19 and transmitting it to others.
As a result, even after receiving vaccine against COVID-19, it’s critical to continue taking simple precautions against the disease. Taking these precautions will help you and others stay safe, and they include:
- Avoiding huge groups of people.
- Keep at least a six-foot physical distance from others.
- With others, stay away from enclosed and poorly ventilated spaces.
- For those who cannot maintain physical distance, a mask or other face protection may be necessary.
- Frequently washing your hands.
- If you’re experiencing symptoms of COVID-19, stay at home and avoid interaction with people.
Busting COVID-19 Vaccine Myths
There are still some misconceptions concerning the safety of COVID-19 vaccinations, despite the fact that numerous studies have found them to be completely safe. Facts can help dispel some of the myths:
- COVID-19 vaccinations do not cause the virus to enter the body: Only very little bits of the coronavirus are used in the vaccines, and even these are unable to spread the virus. Vaccines defend against COVID-19 because they teach your immune system to recognize the virus.
- A person’s DNA is not harmed by the COVID-19 vaccine: Vaccines cannot reach your DNA in the nucleus of your cells because the nucleus is too small to hold them.
- The COVID-19 vaccine has no effect on the ability of women to become pregnant. No studies have revealed any influence on a woman’s ability to conceive due to these or any other immunizations.
- Pregnant women can receive COVID-19 vaccinations without risk. Pregnant women can safely receive vaccinations, according to an increasing body of evidence. Protecting pregnant women from serious illness is another benefit of vaccines. COVID-19. Pregnant women and lactating mothers may benefit from vaccinations throughout pregnancy and breastfeeding, according to certain studies.
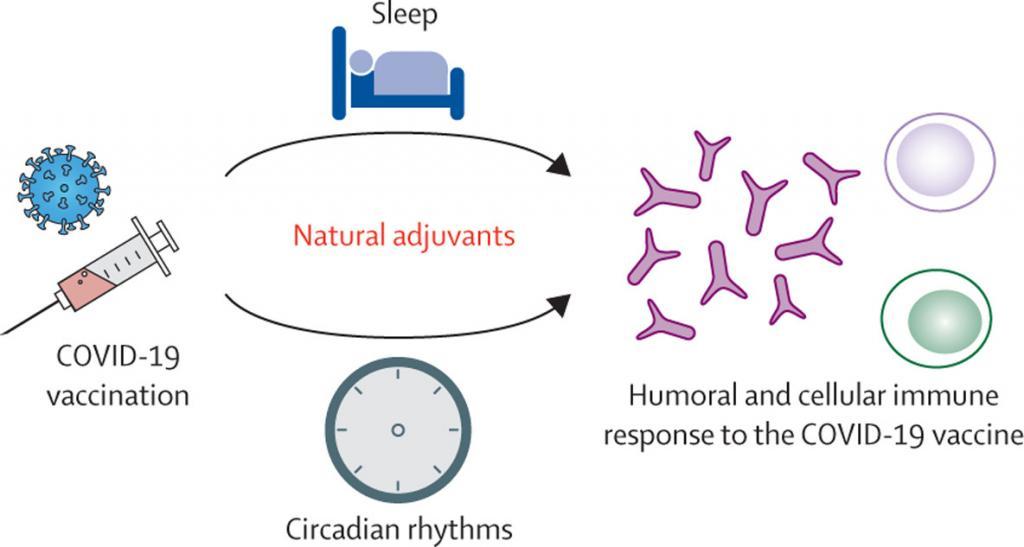
Sleep, Sickness, and Vaccines
Your immune system will benefit if you get a good night’s sleep. Studies on sleep deprivation and other vaccines have indicated that their efficacy can be lowered. However, no research has been done on sleep deprivation and the COVID-19 vaccine.
A good night’s sleep is linked to a lower risk of illness, a better prognosis after an infection, and a better response to vaccines. Contrarily, studies have shown that sleep deprivation reduces the body’s ability to defend itself by reducing the number of white blood cell (immune system cell) travel to the damaged areas of the body.
Our T cells, which are active participants in immunological responses, are another way sleep loss is linked to impaired immune response. Stress hormones are elevated when you don’t get enough sleep, and integrin, a protein that aids T cells in adhering to virus-infected cells, is decreased. As a result of a lack of sleep, T cells may be less effective.
Sleep deprivation impacts the immune system’s reaction to immunizations, especially the flu vaccine. Sleep-deprived patients (those who get less than 7 hours of sleep each night) have a weaker immune response and are more likely to be vulnerable after immunization.
Those with sleep disorders may be concerned about the vaccine’s effect on their current medications or whether it may worsen their current condition. Sleep-wake disorders expert Dr. Michael Thorpy says that “there is no evidence that it is likely to worsen any sleep disorder, and all patients are encouraged to be vaccinated as soon as possible” because of the lack of evidence. As Thorpy points out, sleep apnea is connected with worse outcomes in persons who contract COVID-19, thus vaccinating patients with sleep apnea is critical.
Pfizer and Moderna Vaccinations’ vaccines have “adverse effects that are rare but moderate,” says Thorpy, “such headache, and are generally connected with the second dosage.” Taking acetaminophen after vaccination can help alleviate some of the side effects.
Dr. Thorpy further points out that “better sleep at night is generally related with the satisfaction of being vaccinated,” as he says.
Ongoing COVID-19 Vaccine Research
Research into additional crucial areas of COVID-19 vaccine research is ongoing despite vaccines having already proven significant benefits, such as:
- Existing vaccinations’ ability to protect against newly evolved viral strains.
- Changes to the amount or timing of vaccine doses will have an impact on their effectiveness.
- Some vaccinations’ safety and efficacy in children and teenagers.
- How many people must be immunized in order to provide protection to a larger portion of the population?
For this reason, the FDA and CDC are working with healthcare professionals across the country on several programs to monitor vaccine reactions and immediately identify reports of adverse effects to guarantee safe vaccination programs. Your healthcare practitioner should be contacted if you have any questions or concerns regarding the immunization or any possible side effects.
Get a Good Night’s Sleep Before Your COVID Vaccine
That’s because a healthy immune system is built on a foundation of restorative sleep.
AASM president Dr. Kannan Ramar said in a news release, “As COVID-19 vaccines are delivered, it is of the utmost significance that patients continue to prioritize their sleep in order to preserve good health. “Your immune system is strengthened and your vaccine response is optimized when you get enough, high-quality sleep on a regular basis.”
A number of studies have established a correlation between vaccination response and a person’s ability to sleep. The International Journal of Behavioral Medicine discovered in 2020 that flu vaccines tend to be more effective in persons who obtain adequate sleep the night before receiving the injection. Other studies have found that patients’ responses to hepatitis A and B vaccinations are similar.
At the University of Massachusetts Medical School in Westborough, Dr. Khurshid Khurshid is the director of the UMMHC/UMMS Center for Neuromodulation. According to what he stated, “Sleep has an important function in enhancing the body’s innate and acquired immunological responses. Everyone, but especially health care professionals, should be aware of the positive impact that sleep has on immunity. Immune response to an invading antigen can be boosted by a normal night’s sleep following immunization, and this impact is clinically meaningful.”
“A good night’s sleep before and after immunization could be quite beneficial,” Khurshid wrote in a press statement.
Adults need seven hours of sleep each night to function properly, but the COVID-19 pandemic has caused sleep deprivation in many Americans, according to a new AASM poll.
Thirty percent of those polled indicated their ability to fall asleep has changed, and 29 percent said their nightly sleep has been disrupted as a result.
3 tips for getting a good night’s sleep:
- A bedtime and morning routine should be established. Do not watch TV or read in the bedroom; it is there solely for sleeping. Make sure your bedroom is dark, quiet, and comfortably chilly.
- Turn off your TV and other electronic gadgets 30 minutes to an hour before going to bed to reduce your exposure to blue light. Set your phone on silent and keep it out of reach of your bed so you’re not tempted to check social media or the news while you’re in bed.
- Before going to bed, try to cut back on things like alcohol, caffeine, and heavy meals. Limit yourself to short, sugar-free and easily digestible foods after dinner if you want to prevent interrupting your sleep.
Nguồn: https://www.sleepyheadpillowcase.com
Danh mục: Sleep Advisors















